Dynamic launch mapping
Dynamically maps tasks, dependencies, compliance requirements, and resources across territories so teams can compress timelines with clearer sequencing.

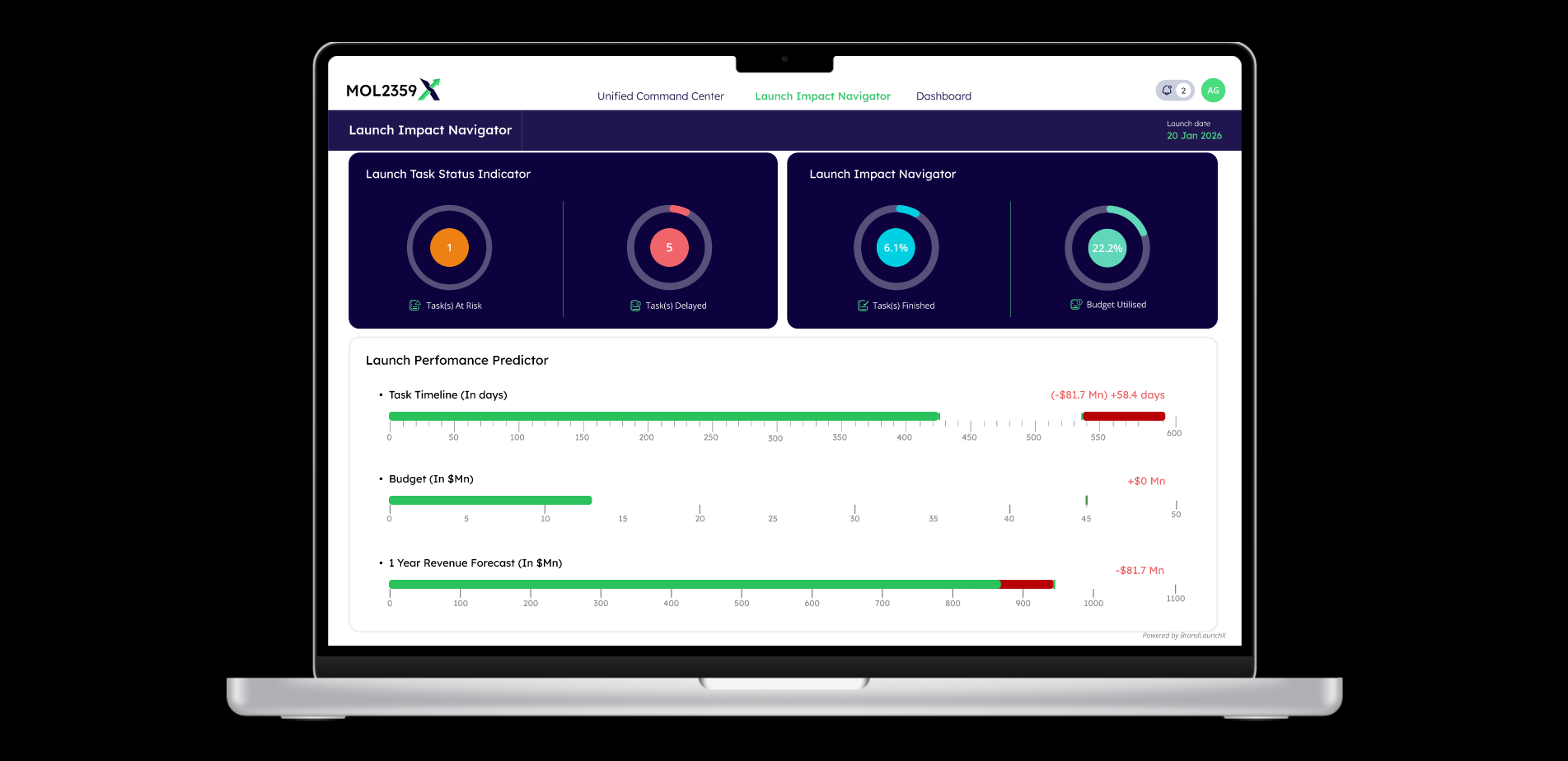
Time to market shapes revenue trajectory, investor sentiment, competitive position, and patient access. BrandLaunchX is an AI-first Launch Impact Navigator and Orchestrator for complex biotech launches that helps teams move faster through critical-path optimization and coordinated decision-making.
The objective is not speed at any cost. It is faster, more disciplined life sciences commercialization with stronger operational stability, clearer accountability, and more reliable launch integrity.
COMMON OPERATING PICTURE. Optimize launch-critical timelines and strengthen collaboration without weakening governance.
END SILOS. Replace fragmented updates with one, robust and coordinated operating view.
CONSTANT GOAL ALLIGNMENT. Keep commercial acceleration tied to earlier patient access.
Emerging risk becomes visible while action is still possible.
Cross-functional leaders align on the same priority stack.
Momentum holds through stage gates instead of stalling between them.
Launch complexity has outpaced the tooling designed to support it. Many organizations still run biotech brand launch execution through static trackers, fragmented reporting, retrospective dashboards, and manual cross-functional reconciliation.
That creates more than administrative friction. It slows decisions, reduces response speed, and leaves commercial value and patient access exposed when the launch window is least forgiving.
Over 80% of new therapeutic launches underperform expectation, and single-day delays can cost up to $16M.
Every delay also slows geographic availability and pushes patients further from access.
Spreadsheet-based launch plans do not recalculate dependencies fast enough when milestones, resources, or territory assumptions change.
Commercial, market access, medical, regulatory, and supply teams often work from different status views and different timing assumptions.
Retrospective dashboards explain what happened. They do not reliably show what will slip next or where intervention matters most.
Launch environments can involve 12 to 16 interdependent teams and 1,000+ concurrent tasks. Manually reconciling that complexity slows decision velocity.
In pharma go-to-market planning, launch speed is a strategic control issue. Teams need a forward-looking operating model, not a better archive of missed signals.
BLX supports earlier visibility, better decision velocity, and more coordinated execution. The platform is forward-looking, not limited to monitoring progress after the fact. It does not replace expertise. It strengthens it.
BLX clarifies which dependencies truly govern launch timing so teams can focus on the work that changes the date, not just the status report.
Teams can assess sequencing choices, resource implications, and territory tradeoffs with more rigor before delay risk becomes embedded.
Coordinated reviews, escalation paths, and decision support help acceleration happen through discipline, not operational shortcuts.
BLX helps teams avoid delay by adjusting priorities, ownership, and response paths as new risks, constraints, and opportunities emerge.
The result is faster cross-functional coordination, earlier intervention, and stronger control across launch readiness, market access, and execution planning.
Critical-path optimization, scenario modelling, delay avoidance, cross-functional coordination, and practical decision support.
Acceleration is delivered with control, not through weakened governance, diluted ownership, or less disciplined launch management.
Each module plays a distinct role in accelerating launch readiness while keeping stage gates, auditability, and regulated-environment readiness explicit. Security and compliance credibility are part of the operating model, not an afterthought.
Dynamically maps tasks, dependencies, compliance requirements, and resources across territories so teams can compress timelines with clearer sequencing.
Identifies emerging risks before they escalate so teams can intervene sooner and avoid avoidable delay in biotech and pharma go-to-market execution.
Simulates remediation scenarios with resource and cost impact analysis so leaders can choose the fastest credible path instead of reacting on instinct alone.
Supports launch blueprinting, access acceleration, change alignment, and structured value reviews so execution stays commercially focused as conditions evolve.
Coordinated stage gates, role-based visibility, and enterprise security controls help faster execution hold up under real regulatory and executive scrutiny.
BrandLaunchX is built around the idea that better launch execution should improve both business performance and patient access. In practice, that means faster visibility, sharper sequencing, and more credible intervention before delays compound.
The messaging framework supports up to 20% reduction in time to market as an early modeled outcome, not a guaranteed claim.
Better commercial outcomes enable better patient outcomes because every week or month saved can bring therapy availability forward.
*Early modelling and market assessment. These figures indicate potential outcome range and should not be interpreted as guarantees.
Six-figure contractual commitments secured
Enterprise pharmaceutical client engagement under non-disclosure agreement.
Good Practice (GxP)-aligned architecture built for regulated life sciences.
Enterprise-grade security, compliance, auditability, and role-based control.
Co-created with pharma brand launch experts and information technology specialists.
BLX helps teams move faster without weakening governance or launch integrity. Use a live discussion or platform walkthrough to assess how the operating model fits your launch environment, timing pressures, and decision structure.
Best for teams with an active timeline, cross-functional complexity, or visible launch risk.
Best for leaders who want a structured look at modules, governance, and workflow logic first.
Commercial acceleration and earlier patient access become easier to pursue together.
Review where launch readiness, market access, and stakeholder decision points create drag.
See how BLX connects signals, governance, and action across life sciences commercialization workflows.
Decide whether to go deeper on platform fit, proof points, or security and compliance review.
Contact us for more details
Learn More